AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Oxygen electrons8/6/2023  
They are also the respective "SPDF" configurations of hydrogen and helium. Since it can have either one or two electrons, its called as $1s^1$ and $1s^2$ respectively. The first shell has only an s orbital, so its called as 1s. The s-subshell can fit 2 electrons, p-subshell can fit a maximum of 6 electrons, d-subshell can fit a maximum of 10 electrons, and f-subshell can fit a maximum of 14 electrons. 
These subshells are called as s, p, d, or f. The number of subshells each shell has depends on the number of the shell (like 1st shell, 2nd shell a.k.a principal quantum number). Its a fact that each shell itself is composed of subshells (experiments involving spectra have shown this). However, that works only up to a certain level. You might have been taught about eletron filling order like: 2 electrons in K shell, 8 electrons in L shell and so on. I will try my best to explain in a way you can understand. But quantum chemistry method will give you mathematical understanding of why there is 2 rooms/apt or why building 2 has 5 apt etc.įirst of all, I must appreciate your eagerness to learn new stuff (though it makes it harder to explain). Now if you want to know more you can read:īut they all are superficial theory, they can say what is going on but they can't say why. Total number of electron/room = 3apt * 2rooms/apt = 6 rooms or 6 electrons Total number of room = 1apt * 2rooms/apt = 2rooms or 2 electrons 0 that means you are living in s orbital. Now, The building name can be rephrased as spdf orbital. Spin quantum number: Each apt has two rooms (room A and room B) (This is actually unrestricted Hartree-Fock case) but if you are living with spouse than you can have a large room by breaking the wall between those two rooms (Only room A or restricted Hartree-Fock case) Magnetic quantum number: This is like apt number on that building. Like for n=1, l=0 (Here l=azimuthal quantum number/building number) but if n=3, then l=0 to (n-1), that means in Sector 3 city there are 3 building named 0,1 & 2. Now if you are living in a city that is very small and has only one building, then you don't need to specify each building of that city differently. It is named as n=1,2,3.Īzimuthal quantum number: This is like the building you live in. By principal quantum number, we actually mean that the probability of finding that electron is high within that particular radius. 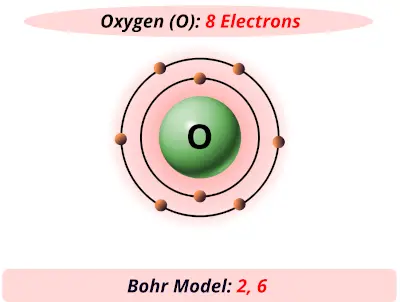
Like sector 1 is the city which has a mean radius of 1 unit and so on. Imagine you are living in a circular states and the cities are named as the radius it has. Now if we want to distinguish between those electrons than we need to name them or there should be something to distinguish. Principal quantum number (This is like the city you live in). Are you familiar with quantum numbers? There are 4 different quantum numbers: Whatever I would like to think about it from a different perspective. Now the basic of this concept is from very fundamental quantum chemistry formulation but I don't think you need to know that now (It is usually taught in 4th year of UG or 1st year of graduate study in Engineering discipline but i am not sure about pure science discipline). By SPDF configuration, he meant orbital configuration. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
